Objective
This study was undertaken to compare the rate of change in cognition between glucocerebrosidase (GBA) mutation carriers and noncarriers with and without subthalamic nucleus deep brain stimulation (STN-DBS) in Parkinson disease.
Methods
Clinical and genetic data from 12 datasets were examined. Global cognition was assessed using the Mattis Dementia Rating Scale (MDRS). Subjects were examined for mutations in GBA and categorized as GBA carriers with or without DBS (GBA+DBS+, GBA+DBS-), and noncarriers with or without DBS (GBA-DBS+, GBA-DBS-). GBA mutation carriers were subcategorized according to mutation severity (risk variant, mild, severe). Linear mixed modeling was used to compare rate of change in MDRS scores over time among the groups according to GBA and DBS status and then according to GBA severity and DBS status.
Results
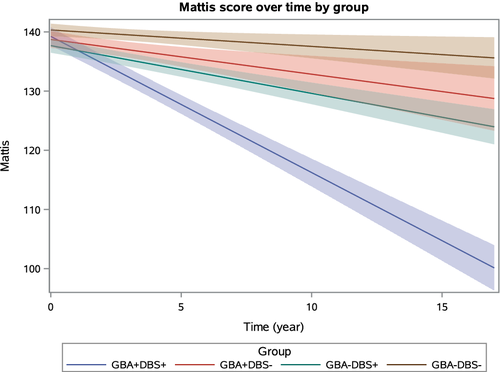
Data were available for 366 subjects (58 GBA+DBS+, 82 GBA+DBS-, 98 GBA-DBS+, and 128 GBA-DBS- subjects), who were longitudinally followed (range = 36–60 months after surgery). Using the MDRS, GBA+DBS+ subjects declined on average 2.02 points/yr more than GBA-DBS- subjects (95% confidence interval [CI] = −2.35 to −1.69), 1.71 points/yr more than GBA+DBS- subjects (95% CI = −2.14 to −1.28), and 1.49 points/yr more than GBA-DBS+ subjects (95% CI = −1.80 to −1.18).
Interpretation
Although not randomized, this composite analysis suggests that the combined effects of GBA mutations and STN-DBS negatively impact cognition. We advise that DBS candidates be screened for GBA mutations as part of the presurgical decision-making process. We advise that GBA mutation carriers be counseled regarding potential risks associated with STN-DBS so that alternative options may be considered. ANN NEUROL 2022
Source: https://onlinelibrary.wiley.com/doi/10.1002/ana.26302